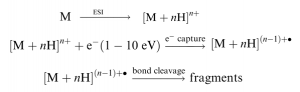- This event has passed.
Chemistry & Biochemistry Colloquium – Roni – Arnold Research Group – Electron Capture Dissociation: A Powerful Technique for Peptide Analysis by Mass Spectrometry
March 9, 2018 @ 2:00 pm - 3:00 pm
Abstract
The structure analysis of large biomolecules such as proteins and peptides by mass spectrometry (MS) is very challenging. However, recently introduced MS techniques facilitate this process by controlled fragmentation. Often physiological modifications take place at the amino acid side chain of proteins such as phosphorylation, sulfation, intra and intermolecular disulfide linkage and glycosylation called posttranslational modification (PTM) to mediate certain cellular functions. Conventional fragmentation techniques of tandem MS such as CID and IRMPD fail to retain these posttranslational modifications while fragmenting the precursor ion during analysis. However, it is very important to identify and locate those PTMs along with the protein structure to understand the biological pathway and action of proteins. We will introduce electron capture and electron transfer dissociation MS that overcome earlier limitation of MS for protein structure analysis and PTM identification. This technique produces multiple charged precursor ions by ESI and exposes them to low energy electrons. Through this controlled process, ions become charge reduced and fragment into c and z ions.
Reference:
- Roman A. Zubarev, Neil L. Kelleher, and Fred W. McLafferty, J. Am. Chem. Soc. 1998, 120, 3265-3266
- Konstantin O. Zhurov, Luca Fornelli, Matthew D. Wodrich, Unige A. Laskay and Yury O. Tsybin, Chem. Soc. Rev., 2013, 42,5014–5030
- Scott R. Cole, Xiaoxiao Ma, Xinrong Zhang, Yu Xia, J. Am. Soc. Mass Spectrom. (2012) 23:310Y320
- Mirela Sarbu, Roxana M. Ghiulai, Alina D. ZamfirAmino Acids (2014) 46:1625–1634 DOI 10.1007/s00726-014-1726-y
- Yulin Qi and Dietrich A. Volmer, Analyst, 2016,141, 794–806
- Nicholas M. Riley and Joshua J. Coon, Anal. Chem. 2018, 90, 40−64